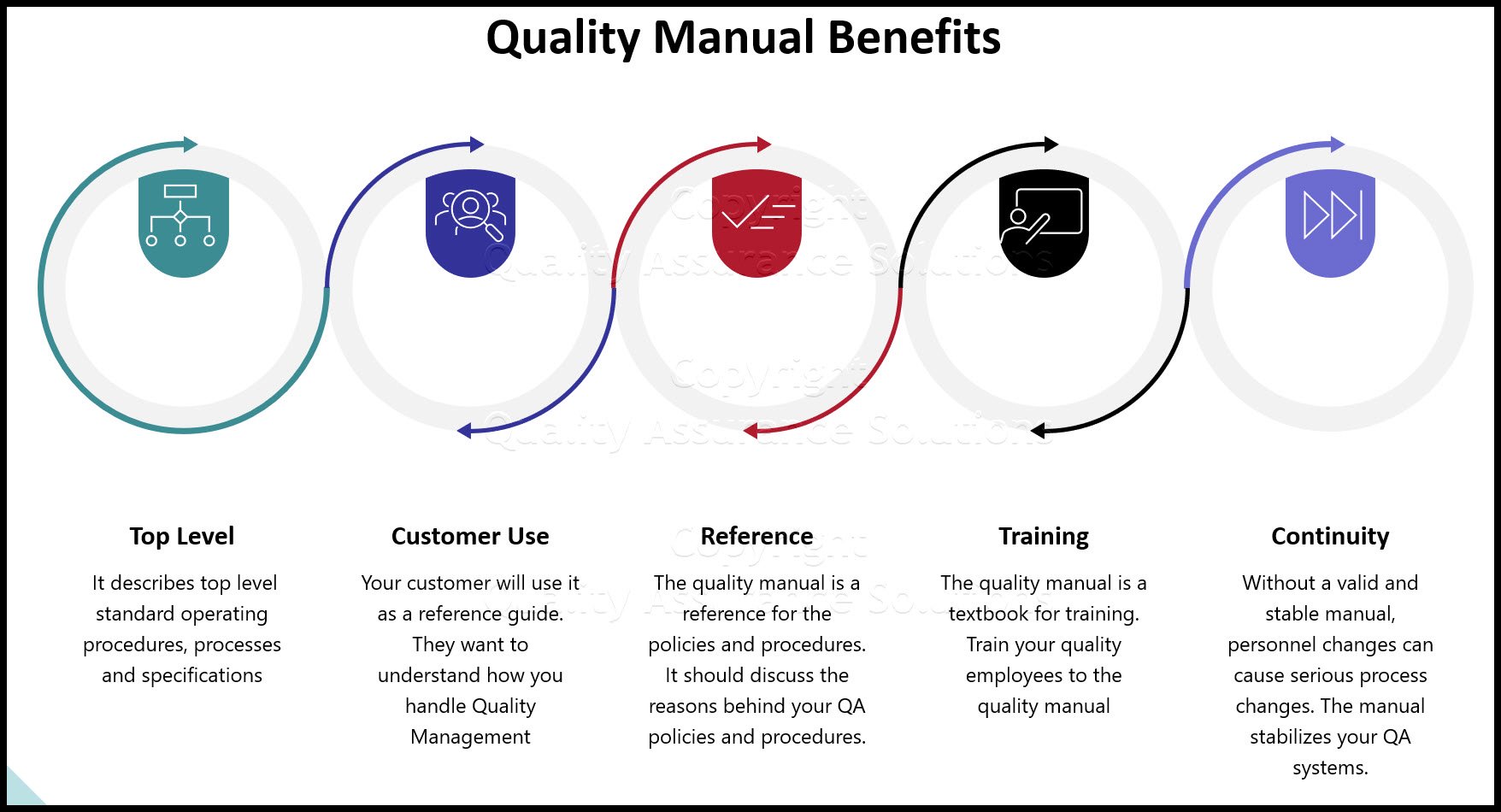
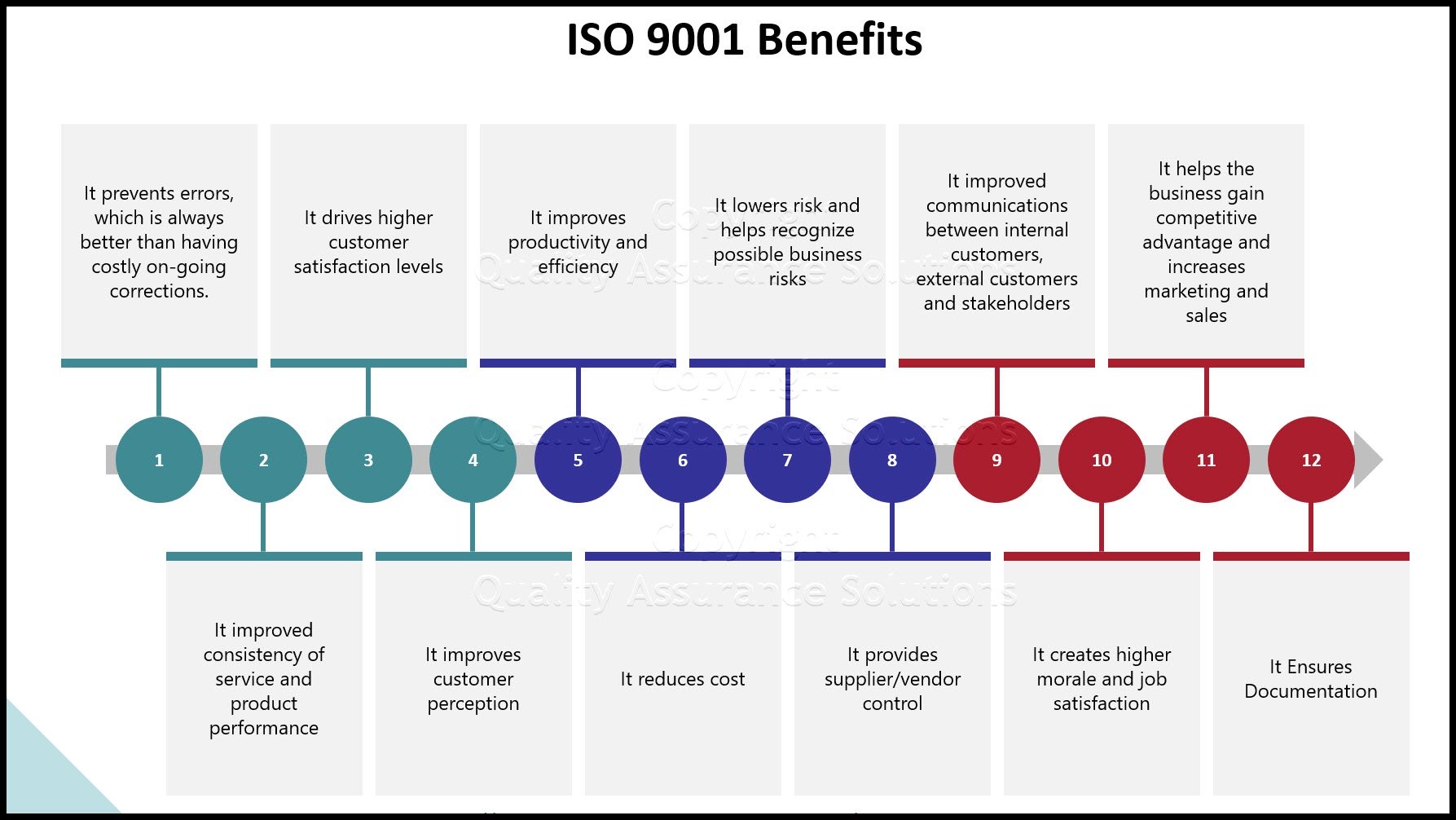
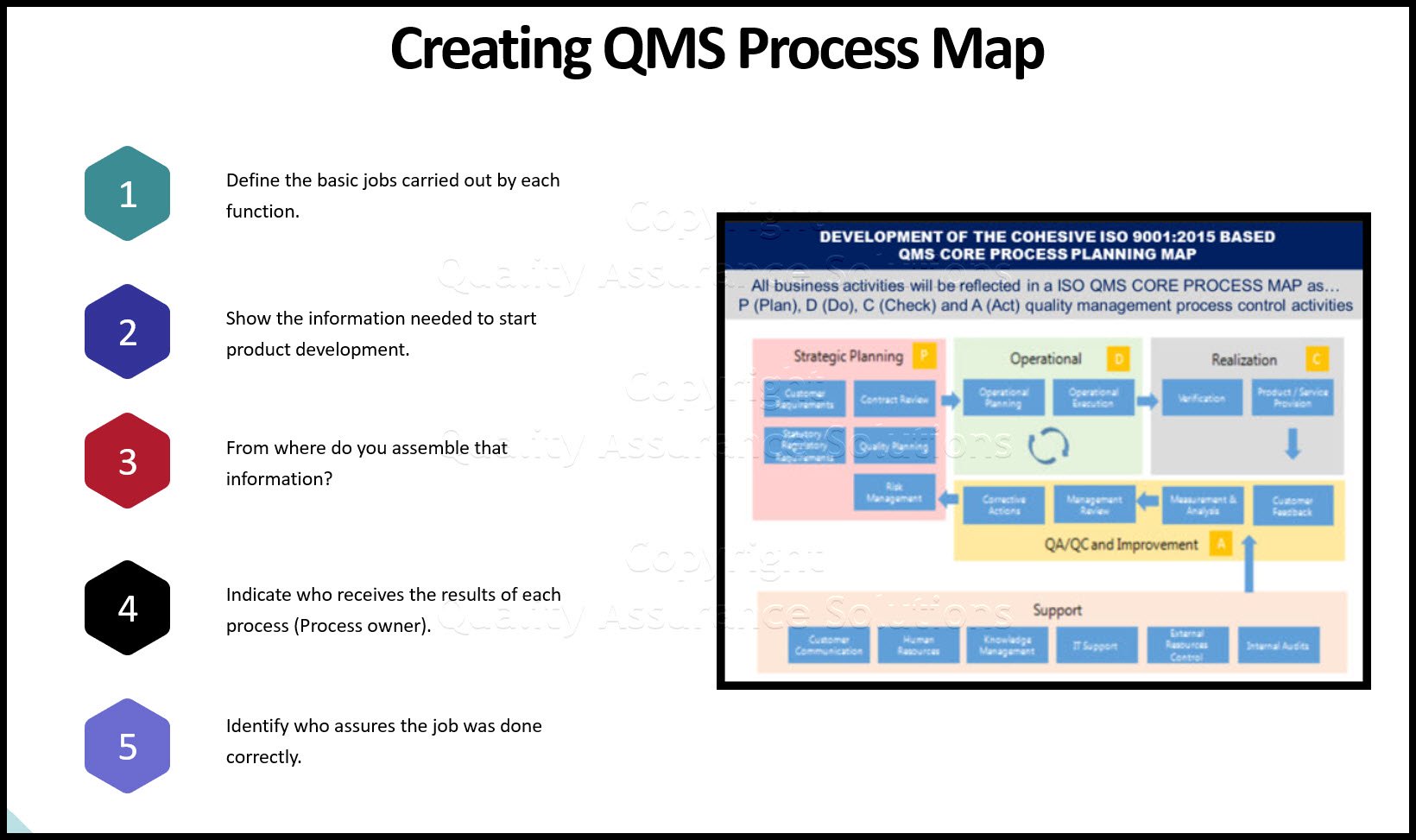
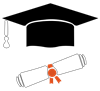
Quality Program
Management Review
The Quality Program Management Review is critical and necessary for ISO 9001 certification. Not only this, but when done properly, it drives extensive improvement throughout your business. In theory, the QMS review output creates the plan for implementing, changing and improving your quality program.
During your ISO 9001 certification and monitoring audits, the auditor always examines your quality management review quality records.
Your easy to edit ISO 9001:2015 Quality Assurance Manual.
QMS Review Frequency:
According to the ISO 9001 standard, the quality management system must be reviewed on a periodic basis. There are a couple of approaches to meeting this requirement.
- Annual Schedule Meeting
- Semi Annual Meeting
- Weekly Meeting
If you’re starting to implement the ISO 9001 system then the review should be a semi annual meeting. Meet twice a year until your system matures. This assures your company stays on track and does not lose focus from the standard.
If your company has a mature system and does not expect many changes to the QMS operation then conduct an annual meeting.
Instead of an annual or semi annual meeting, a management weekly meeting could be used. When taking this approach, track all input and output records. During these meetings cover the inputs for the Quality Program Management Review and record the action items. You do not need to cover all input requirements during each meeting, however you must prove (provide documentation) that you do access the input requirements on a regular basis.
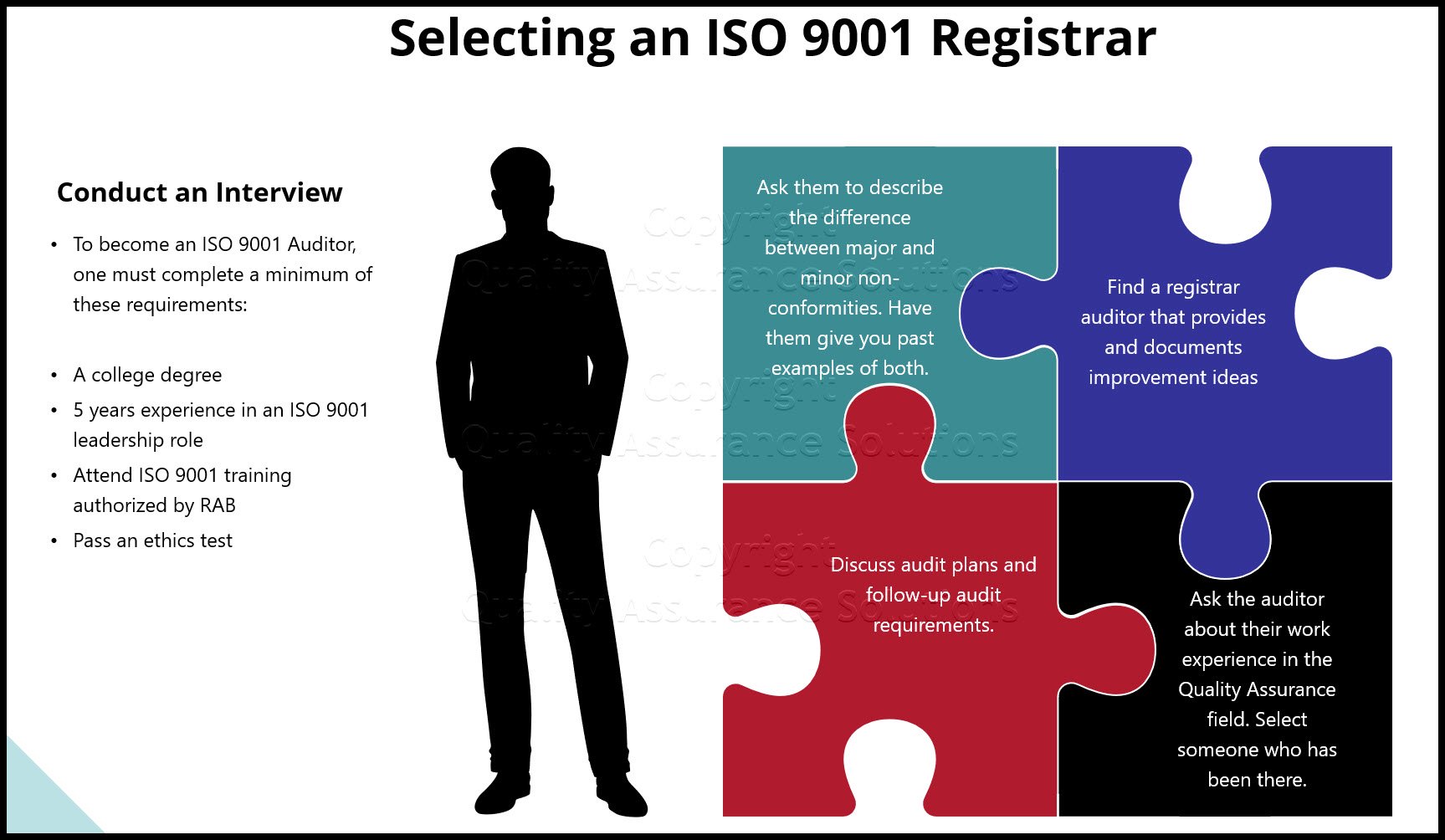
Quality Program Management Review Procedure
I recommend creating a procedure that documents the management review process. Within the procedure discuss the frequency, the required management team, the mandatory inputs, the outputs and your quality records. This makes it easier for you to assure you meet the requirement.
Note, this procedure is not absolutely necessary for ISO certification. See here for a list of required procedures. If you choose not to maintain a procedure, then be sure to keep your quality records for your auditors evaluation.
Your easy to edit ISO 9001:2015 Quality Assurance Manual.
Quality Program Management Review Team
When the ISO 9001 standard uses the word “Review”, this means that “a team analyses the inputs and the team makes the output plan”. For the review, employ a cross functional team approach.
When selecting the team members, include your senior management. I recommend taking a look at your organizational chart and select the top 2 or 3 management levels for your given location. The top 2 or 3 tiers are the people who belong on the quality management review team. Include managers of these functions during the QMS review meeting:
- President or Operations Manager
- Quality Assurance Manager
- Engineer Manager
- Facilities Manager
- Production Manager
- Purchasing Manager
- Sales Manager
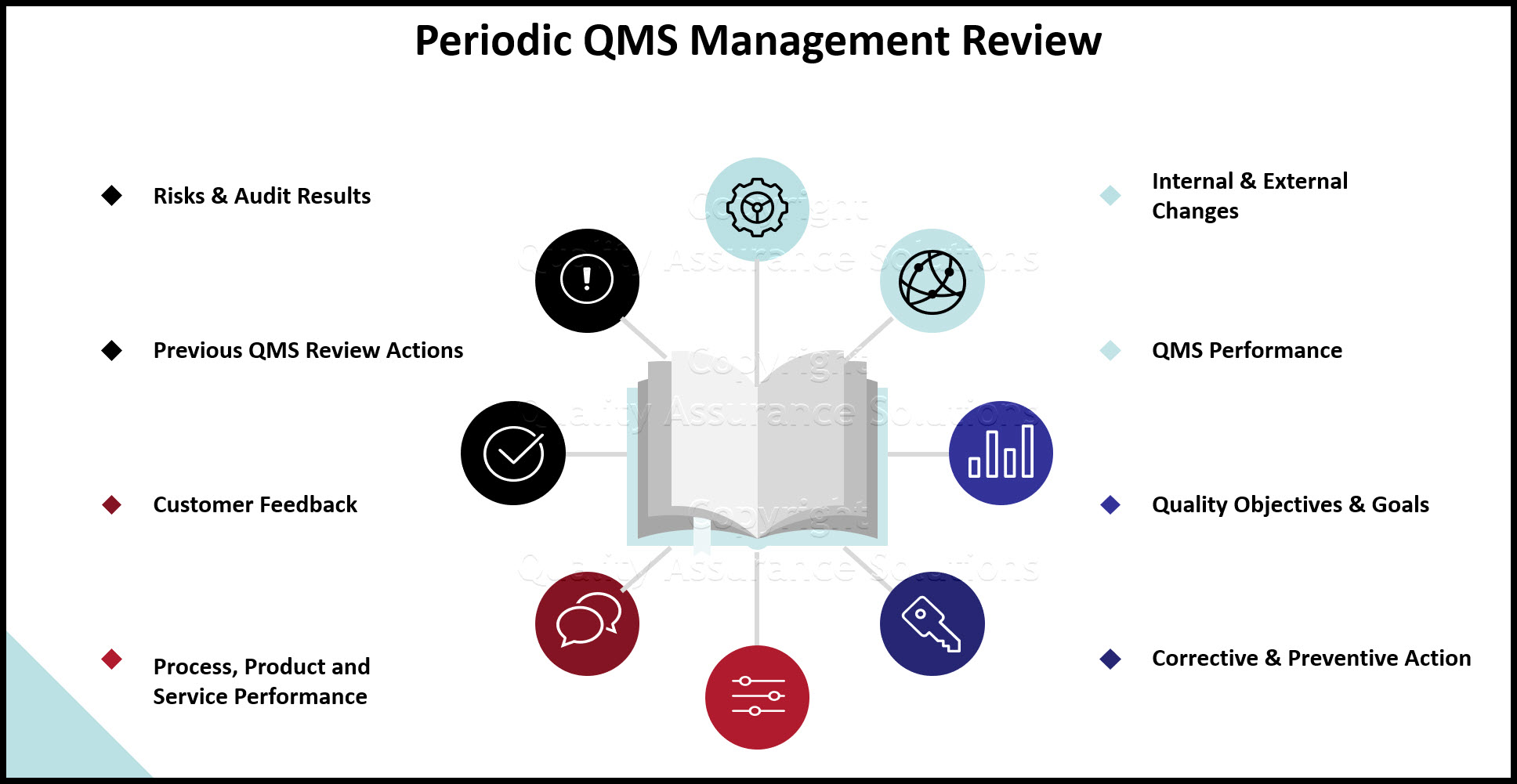
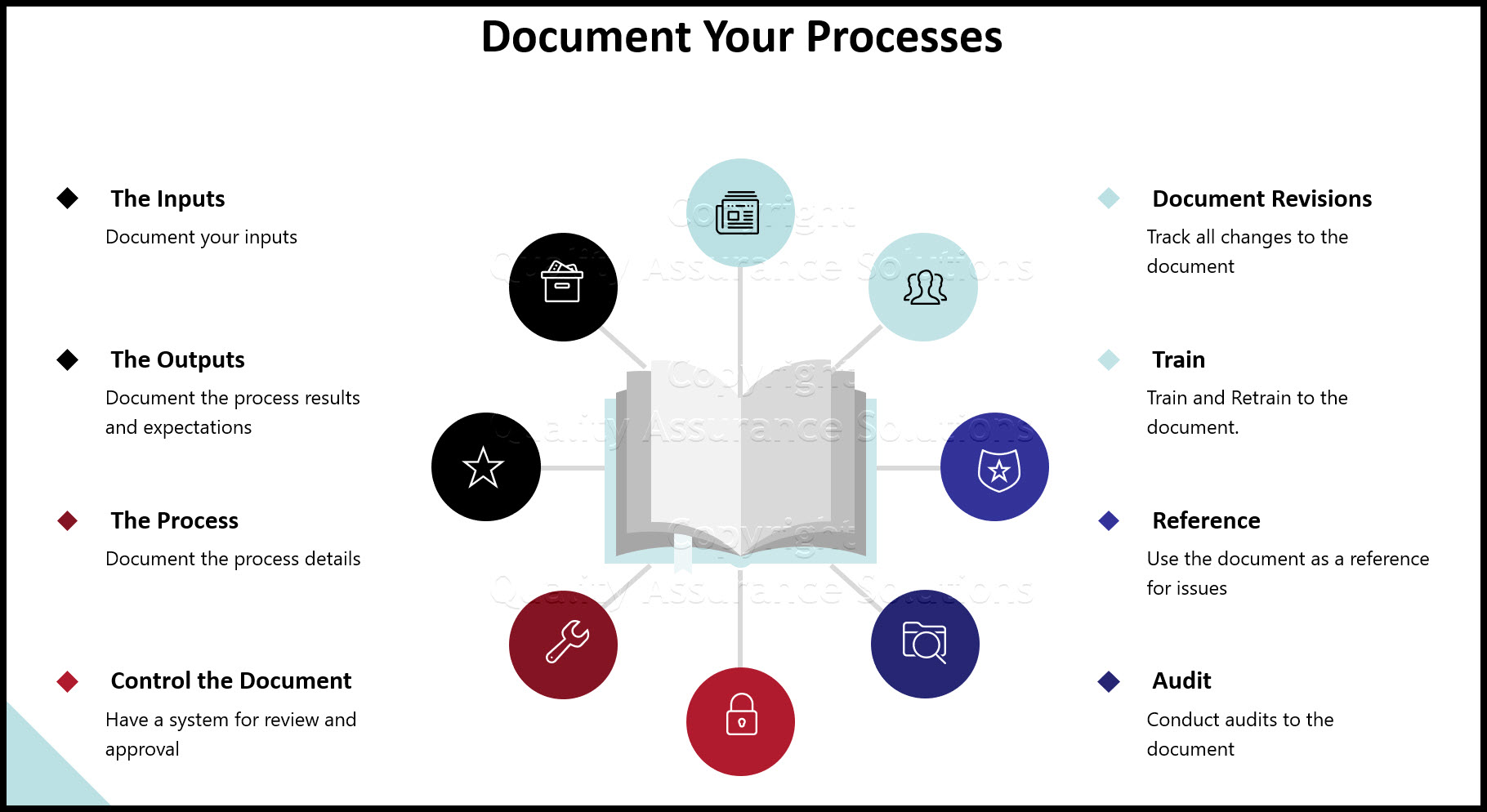
QMS Review Inputs
The team discusses and evaluates the inputs during the quality program management review. The inputs are actual quality records which accumulated since the last management review meeting. Below is a list of necessary inputs:
- The output (plan) from the previous QMS Review
- customer feedback
- process performance data
- product conformance data
- quality metrics
- quality policy
- Key process areas from the interaction chart
- corrective actions
- changes that could affect your quality management system
- Improvement recommendations
- risk management issues
Your easy to edit ISO 9001:2015 Quality Assurance Manual.
QMS Review Outputs
The output is a list of action items that addresses the inputs. Document this on a gantt chart or a to do list. For each action item, include the following
- action item or task
- person responsible
- due date
- state if it is an improvement, preventive or corrective action measure.
On the final report, collect signatures of each team member and record the date of review.
QMS Documentation
Collect all the input data and the final output report and place this information into a folder or binder. Label the binder with “QMS review and the date.” Your ISO auditor will assess this package.
ISO 9001 Audit
The auditor will ask you for the Quality Program Management Review documentation. Present the package and let him assess it. Be sure to meet your output action items. He will ask for you to provide status on the action items. The auditor will also quiz you on your inputs. He wants to know why you selected the given inputs. He wants to know if you addressed problematic inputs. In addition, the auditor will quiz the members of the review team. He wants to make sure the quality management review was a team effort.
See this list of key elements for a QA Project Plan
Key Elements for a Project Quality Management Plan
- QAS Home
- QMS Review
|
Quality Assurance Solutions Robert Broughton (805) 419-3344 USA |
 |
|
Software, Videos, Manuals, On-Line Certifications | ||
|
An Organizational Task Management System. Projects, Meetings, Audits & more | ||
|
Corrective Action Software | ||
|
Plan and Track Training | ||
|
AQL Inspection Software |
|
450+ Editable Slides with support links | ||
|
Learn and Train TRIZ | ||
|
Editable Template | ||
|
Templates, Guides, QA Manual, Audit Checklists | ||
|
EMS Manual, Procedures, Forms, Examples, Audits, Videos | ||
|
On-Line Accredited Certifications Six Sigma, Risk Management, SCRUM | ||
|
Software, Videos, Manuals, On-Line Certifications |